Physical Representations
|
Type
pH range
|
xlogP
|
Des A‑Pol
Apolar desolvation
(kcal/mol)
|
Des Pol
Polar desolvation
(kcal/mol)
|
H Don
H-bond donors
|
H Acc
H-bond acceptors
|
Chg
Net charge
|
tPSA
(Ų)
|
MWT
Molecular weight
(g/mol)
|
RB
Rotatable bonds
|
DL |
|
Ref
Reference (pH 7)
|
2.65 |
5.25 |
-14.03 |
1 |
6 |
0 |
66 |
439.006 |
4 |
↓
|
|
Mid
Mid (pH 6-8)
|
2.65 |
8.54 |
-49.2 |
2 |
6 |
1 |
67 |
440.014 |
4 |
↓
|
Clustered Target Annotations
| Code |
Organism Class |
Affinity (nM) |
LE (kcal/mol/atom) |
Type |
|
FA10-1-E |
Coagulation Factor X (cluster #1 Of 2), Eukaryotic |
Eukaryotes |
6830 |
0.26 |
Binding ≤ 10μM
|
ChEMBL Target Annotations
| Uniprot |
Swissprot |
Affinity (nM) |
LE (kcal/mol/atom) |
Type |
|
FA10_HUMAN |
P00742
|
Coagulation Factor X, Human |
6830 |
0.26 |
Binding ≤ 10μM
|
Reactome Annotations from Targets (via Uniprot)
| Description |
Species |
|
Common Pathway |
|
|
Extrinsic Pathway |
|
|
Gamma-carboxylation of protein precursors |
|
|
Intrinsic Pathway |
|
|
Removal of aminoterminal propeptides from gamma-carboxylated proteins |
|
|
Transport of gamma-carboxylated protein precursors from the endoplasmic reticulu |
|
Rings
-
Thiazole
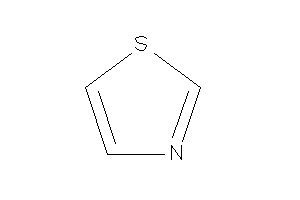
-
Thiophene
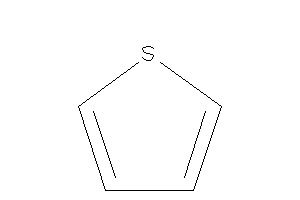
-
1,2,3,6-tetrahydropyridine
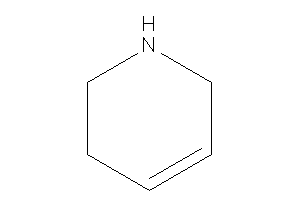
-
Piperidine
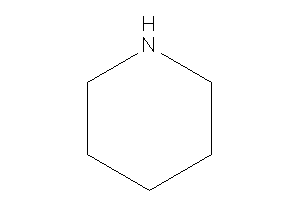
-
4,5,6,7-tetrahydrothiazolo[5,4-c…
![4,5,6,7-tetrahydrothiazolo[5,4-c]pyridine](//zinc12.docking.org/img/rings/4740.gif)
-
N-[[1-(4,5,6,7-tetrahydrothiazol…
![N-[[1-(4,5,6,7-tetrahydrothiazolo[5,4-c]pyridine-2-carbonyl)-3-piperidyl]methyl]thiophene-2-carboxamide](//zinc12.docking.org/img/rings/469005.gif)
No pre-computed analogs available. Try a structural similarity search.